

Interfyl is a decellularized human placental connective tissue matrix (CTM) to be used for the replacement or supplementation of damaged or inadequate integumental tissue.
For more information please contact Celularity at 1-844-963-2273
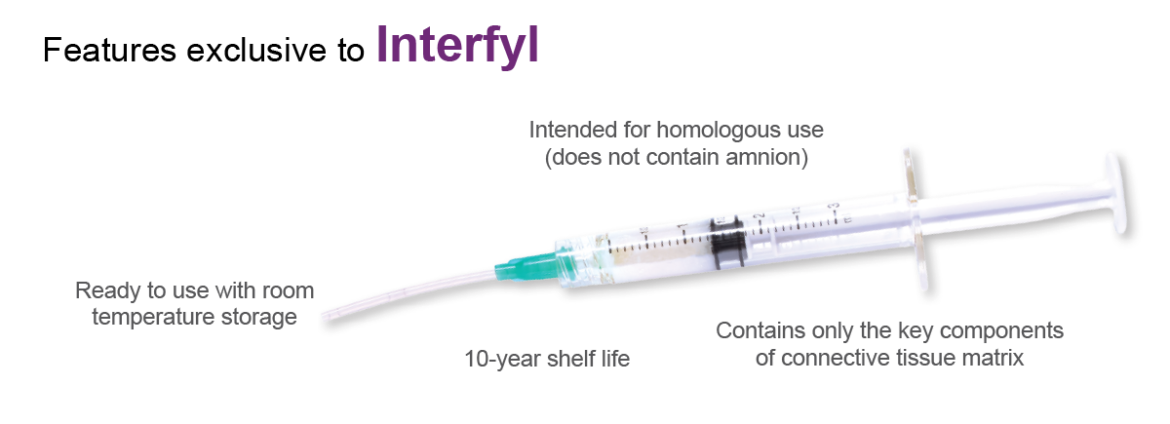
Available in flowable and particulate form in units of weight ranging from 45mg to 275mg to meet a variety of application needs

1.5 mL Flowable (275mg)
Product code: HCTM015
1 mL Flowable (170mg)
Product code: HCTM010
100 mg Flowable
Product code: HCTM001
0.6 mL Flowable (75mg)
Product code: HCTM060
0.3 mL Flowable(45mg)
Product code: HCTM030

50 mg Particulate
Product code: HCTM050
100 mg Particulate
Product code: HCTM100
WE PUT SAFETY AT THE TOP OF THE LIST
Interfyl® tissue procurement and processing practices meet high safety standards.
Tissue used in processing Interfyl:
- Has been procured, processed, and tested in accordance with standards established by the American Association of Blood Banks (AABB) and the United States Food and Drug Administration (FDA)
- Passed safety testing for cytotoxicity, hemolysis, irritation, endotoxins, and pyrogenicity.
- Utilizes a barcode tracking system for optimal safety monitoring and to enhance patient and practitioner confidence.
Certifications
Celularity adheres to high standards of ethical practice and transparency in all aspects of our operations. Celularity is registered with the FDA as a tissue establishment and accredited by the AABB.
Indications for Use
lnterfyl is intended for use as the replacement or supplementation of damaged or inadequate integumental tissue.
Contraindications, Warnings, and Precautions
Interfyl is contraindicated in patients with a known hyper-sensitivity to Interfyl.
Interfyl should not be used in clinically infected sites. If a recipient had an adverse reaction related to previous use of Interfyl, do not re-apply.
Do not use Interfyl for intravenous, intra-arterial, intra-ocular, or intrathecal applications.
Interfyl must be used prior to the expiration date on the product pouch. Once opened, Interfyl must be used within two hours or discarded per institutional procedures. The contents are sterile if the vial/syringe (container) is unopened and undamaged. Do not sterilize.
For product information, product complaints, or adverse reaction reporting, call 1-844-963-2273.
Please refer to the Interfyl Package Insert for complete product information.

Interfyl® and Celularity® are registered trademarks of Celularity Inc.
© 2023 Celularity Inc.